|
4/23/2024 0 Comments Why are entropies absolute  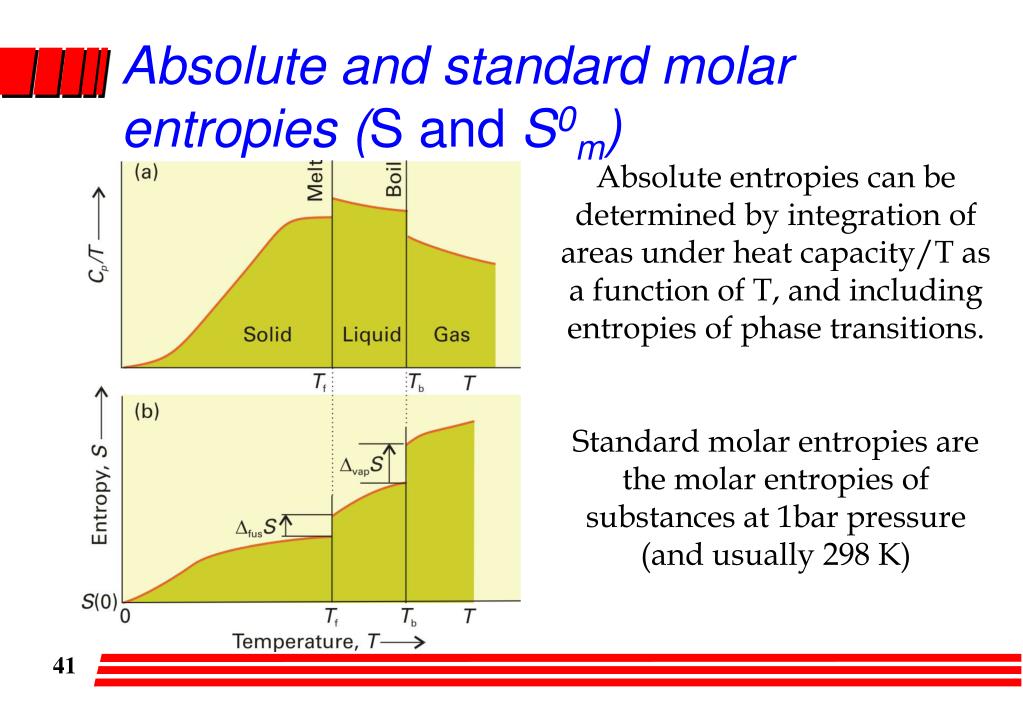
Furthermore, we show some application examples for the calculation of free energy differences in typical chemical reactions. Comprehensive tests indicate a relatively strong variation of the conformational entropy on the underlying level of theory for typical drug molecules, inferring the complex potential energy surfaces as the main source of error. Even for the hardship case of extremely flexible linear alkanes (C 14H 30–C 16H 34), errors are only about 3 cal mol −1 K −1. Extensive tests of the protocol with the two standard DFT approaches B97-3c and B3LYP-D3 reveal an unprecedented accuracy with mean deviations <1 cal mol −1 K −1 (about <1–2%) for the total gas phase molecular entropy of medium-sized molecules. For the first time, variations of the ro-vibrational entropy over the CE are consistently accounted-for through a Boltzmann-population average. 
Anharmonic effects are included through the modified rigid-rotor-harmonic-oscillator (msRRHO) approximation and the Gibbs–Shannon formula for extensive conformer ensembles (CEs), which are generated by a metadynamics search algorithm and are extrapolated to completeness. The scheme is systematically expandable and can be integrated seamlessly with continuum-solvation models. We propose a fully-automated composite scheme for the accurate and numerically stable calculation of molecular entropies by efficiently combining density-functional theory (DFT), semi-empirical methods (SQM), and force-field (FF) approximations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
